White Powecom KN95 - 10 count / bag
- Regular price
- $10.69
- Sale price
- $10.69
- Regular price
-
- Unit price
- /per
Adding product to your cart
Made to the NEW GB2626-2019 Standard with Better Breathability
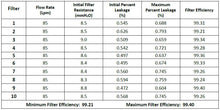
Powecom KN95 respirators are equivalent or higher than the N95 NIOSH rated particulate filtration efficiency.
High Filtration / Industrial / Commercial / Home Use
Dr Terry Dubrow of E! Entertainment show Botched Approves!
SKU: KN95-RSP-PWCM-MSK-10
UPC: 6955984500159
Availability: In Stock
CDC Tested: Yes, See Details Below
Manufacturer: Guangzhou Powecom Labor Insurance Supplies Co., LTD.
Lead/Fulfillment Time: 1-2 Days
Inventory Status: In Stock - Ships From San Mateo, CA - USA
The Powecom KN95 personal respirator ships directly from our San Mateo, California warehouse. Face masks made with a multi-layer filtration system of non-woven breathable fibers that is utilized globally. These masks are manufactured by Guangzhou Powecom Labor Insurance Supplies Co., LTD.
MATERIAL:
46% non-woven polypropelene, 28% melt-blown fabric, 26% ES hot air cotton.
Ear loops: Latex free
Nose piece: Free forming adj aluminum
FEATURES:
Construction: Multi-layer filtration system of non-woven soft and breathable fibers.
All respirators manufactured after May 20th are affixed with an anti-fake sticker that can be used to verify authenticity.
For Guangzhou Powecom Labor Insurance Supplies Co., LTD. KN95 CE certificate, please check Gallery.
Additional Detail
Frequently Asked Questions
KN95 masks initially originated as a counterpart to the N95 respirator. They tend to provide protection close to that of an N95 respirator. While an N95 respirator has straps that go around the head for ensuring a tight seal, a Powecom KN95 face mask has ear loops and does not form a tight seal.
How do I ensure that my Powecom KN95 face masks are not counterfeit?
No matter how cautious you are during your purchase, most masks tend to be counterfeits. So, you need to pay attention to the red flags mentioned below:
-No markings at all
-An N95 that has ear loops rather than head straps
-A statement that a KN95 mask is CDC or NIOSH approved.
-Either no NIOSH stamp or incorrect spelling of NIOSH
Do all KN05 masks need to be fit-tested to ensure maximum protection?
No. Powecom KN95 face masks are not at all suitable for fit-testing. As these masks have not been certified by the National Institute for Occupational Safety and Health, the OSHA does not consider it a negative-pressure respirators.
Do you have any tips for safely using surgical or cloth masks?
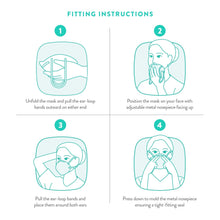
One of the most crucial tips we have for you is to ensure your mask is tightly fitted to your face. Here are a few additional tips for putting on your mask.
Washing or sanitizing your hands immediately before and after putting your mask on, as well as taking it off.
Place the mask on your face in such a way that the moldable nose wire sets against the bridge of your nose and the bottom of the mask lies below your chin.
Ensure that the mask’s straps are placed around each of your ears.
Mold the top of the Powecom KN95 mask to fit the contour of your nose and use both hands to adjust it.
You can even reduce airflow around the mask by adjusting it as you feel like.
Should I wear a cloth mask with my Powecom KN95 face masks?
No. It is advised not to wear cloth masks in addition to a KN95 as it reduces the effectiveness of the surgical mask. You don’t need any extra protection against particulate matter when you are wearing KN95 masks. They are the most effective solution.
Could I know more about your shipping policy?
We ship standard shipping on all domestic orders unless stated otherwise. For expedited shipping, please contact support@planethalo.com.
When do I discard my mask?
You should replace your mask immediately if it:
Becomes wet or soiled in any way
Fails to cover your mouth or nose anymore
Has been stretched out to a point where it’s no longer effective
Has damaged ties or straps
Has trouble staying on your face anymore
Has holes or tears
Do Powecom KN95 face masks have a shelf life?
The shelf life of disposable masks, such as KN95 and surgical masks, is typically two to three years. However, how you keep them can change how long they last. For instance, they could deteriorate more quickly if you expose them to extreme heat and humidity than if you store them at a moderate temperature.